Virus and air humidity #02
In our second article about the effects of air humidity on the spread of virus, we take a look at how air humidity affects the defense mechanisms in the body.
Specifically, we will take a look at a scientific study done on mice exposed to virus – and how low air humidity made the mice much more susceptible to virus infection. This study more than hints that the low indoor air humidity we have in our homes, schools, offices and hospitals during cold months may make it much easier for a virus to infect us.
Many factors affect the spread of virus
Air humidity is of course not the only factor in containing the spread of virus. Factors such as temperature, air pressure, human behavior and much else besides are equally important. But air humidity has often been overlooked as a mitigating factor in the spread of virus, which is a shame, as it is actually something we can control in our indoor environment.
In the first article in this series, we took a look at how air humidity in the optimum range for human comfort – from approx. 40 to 60 % relative – also has the benefit of making it harder for virus to remain airborne for long. And, since flu viruses mainly infect by coming into contact with our respiratory systems, this may be something to consider in the design of our indoor environments.
How we protect ourselves from virus
But normal human activity means that we cannot avoid getting pathogens such as virus into our noses and mouths. What happens then is highly complex, but a healthy body has several powerful defense mechanisms to stop virus and bacteria from doing us harm. In fact, it is only a tiny fraction of the virus we encounter that slip through the defenses – the rest is rendered harmless without us even knowing.
MCC: A conveyor belt for pathogens
MCC: A conveyor belt for pathogens
In our noses and mouths, the first line of defense is the mucosal barriers in the respiratory system. A layer of mucus covers the tissue and prevents virus from getting in contact with the body’s cells.
Secondly, in our noses, mouths and lungs, we have epithelial cells that form the outer surface, the border where our bodies meet the environment, so to speak. Our skin is a form of epithelial layer, as well, protecting us from infection. The types of epithelial cells we have in our respiratory system are covered in a layer of fluids and are also fitted with cilia, tiny hair-like structures that transport the mucus and, with it, trapped pathogens out of the body.
The combined effect of the mucus and the cilia is known as mucociliary clearance (MCC).
 MCC: A conveyor belt for pathogens
MCC: A conveyor belt for pathogens
Tissue repair
After having encountered virus and combatted it, the cilia and the cells are damaged, but have the ability to repair themselves quite quickly; however, while the tissue is damaged, this leaves an opening through which a virus can invade a cell.
Once the virus is in the cell, however, it’s not home free – fortunately, the body and the cells themselves have all sorts of sophisticated defensive mechanisms that are not relevant to this blog post. It’s tough being a virus, but nevertheless, some of them make it through – and make us ill.
How air humidity affects our defenses against virus
Several studies have shown that these defense mechanisms are directly affected by the humidity of the air around us. Low air humidity can make the defense much less effective.
We all know how dry air can make our eyes feel dry and make us feel more thirsty; this is due to the surface tissue in our eyes, noses and mouths losing vapor to the surrounding air. And there are indications that dry surface tissue is much more sensitive to virus infection.
Lab mice get sicker in low humidity
In a recent study using laboratory mice1), three important findings were made:
- That mice infected with influenza virus type A would develop more severe symptoms and have higher lethality when the mice were kept in low relative air humidity (10 – 20%)
- That low relative humidity (10 – 20%) has a negative impact on mucociliary clearance (MCC), impacting both the speed of the MCC and its direction
- That low air humidity makes it harder for epithelial cells to repair themselves after a virus infection
The study made other findings, as well, all of whom point to low air humidity having a negative effect on the body’s ability to protect itself against virus infections.
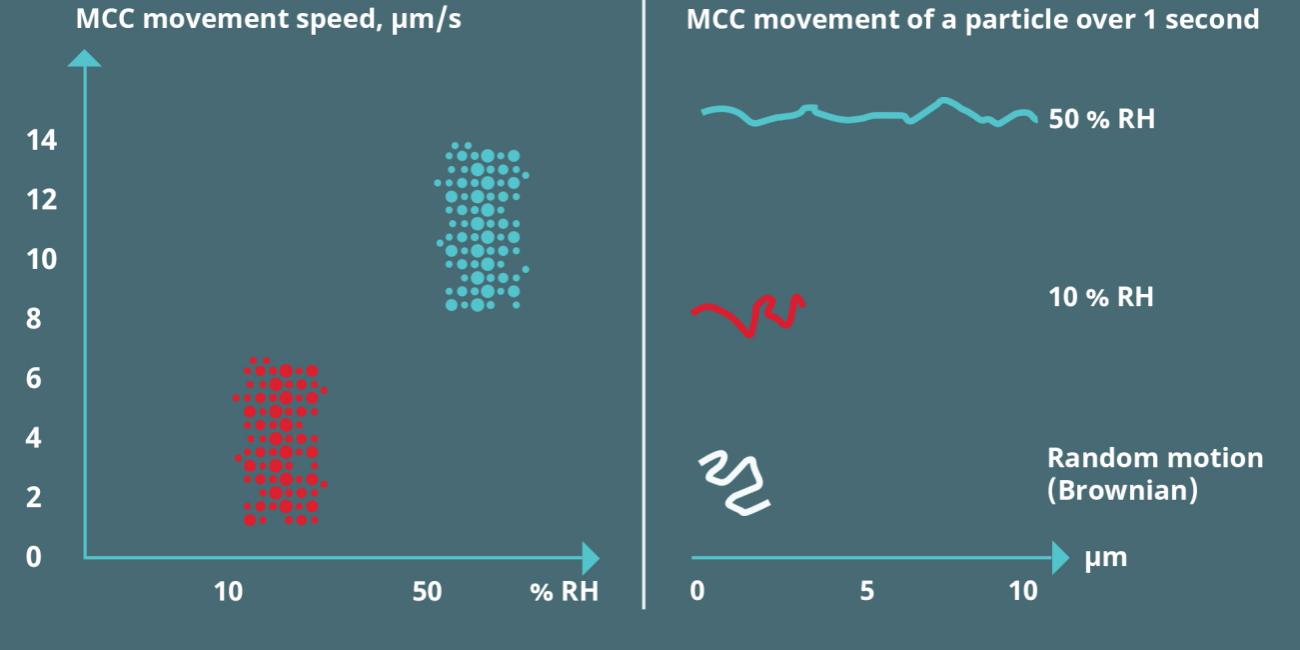
MCC movement and RH. After https://www.pnas.org/content/116/22/10905
You can read the whole paper for yourself and see its results by following the link at the end of this article.
What this may mean
The results are not conclusive and do not suggest that air humidity is the only remedy for the spread of virus-related diseases. Scientists still lack a complete understanding of why flu-related diseases spread in tropical conditions with high humidity, but it is quite clear that there is a link between low indoor humidity in temperature climates and the risk of flu infection.
Air humidity control can be part of the solution to containing virus spread
At Airtec®, we find it interesting to see how study after study point to the positive effects of air humidification. We have developed air humidification systems for decades in order to mitigate the effects related to low air humidity in commercial buildings and industrial facilities: Airborne dust, static discharges, desiccation of products and raw materials, and the removal of excess heat generated by industrial processes. Our systems are also used to create a more pleasant and productive indoor environment for e.g. office workers, museum guests and so forth.
We will continue to follow the scientific developments in the field of air humidity and the spread of virus, and in the next blog post we will take a look at what researchers have to say about the relationship between air humidity and the viability of virus – that is, how low air humidity can possibly make virus stay active and contagious for longer.
Stay tuned for the next one!
Contact us today and find out more
1) Low ambient humidity impairs barrier function and innate resistance against influenza infection
Sources
Eriko Kudo, Eric Song, Laura J. Yockey, Tasfia Rakib, Patrick W. Wong, Robert J. Homer, and Akiko Iwasaki
PNAS May 28, 2019 116 (22) 10905-10910; first published May 13, 2019 https://doi.org/10.1073/pnas.1902840116
Contributed by Akiko Iwasaki, April 4, 2019 (sent for review February 19, 2019; reviewed by Gabriel Núñez and Peter Palese)